We are pleased to share our latest open-access article titled “Acidity scale in 1,2-difluorobenzene” by John Paulo Samin, Helerin Roomet, Märt Lõkov, Sofja Tshepelevitsh, Jaan Saame, Agnes Heering, and Ivo Leito, published in ACS Omega (DOI: 10.1021/acsomega.6c02089).
In this work, we established equilibrium Brønsted acidity measurements in 1,2-difluorobenzene (1,2-DFB) in terms of pKa values measured by UV-vis spectrophotometry and unified pH (pHabs) values measured by differential potentiometry.
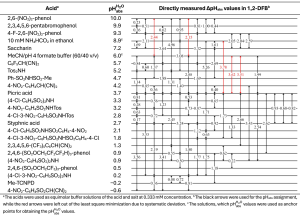
The pKa scale in 1,2-DFB spans 15 orders of magnitude. The pKa values were determined for 137 acids, 33 of which were directly measured (see Table 1). The remaining values were estimated using correlation equations relating pKa values in other solvents (acetonitrile and 1,2-dichloroethane) to those in 1,2-DFB. The pKa scale was anchored to the computational pKa values of 9 acid compounds.
Furthermore, an experimental unified pH scale spanning more than 10 orders of magnitude was established. The scale was aligned to the aqueous pH scale, assigning pHabsH2O values to the buffer solutions of 22 acids ranging from −0.6 to 10.0 (see Table 2). The potentiometrically measured pHabsH2O values showed good agreement with the values independently calculated from experimental pKa values anchored to computational reference values, demonstrating the solid foundation of the underlying theory.
These results open the possibilities for quantitatively describing and measuring acid−base processes in 1,2-DFB, including potentiometric and UV−vis spectrophotometric pHabsH2O measurement.
(This research was supported by grant PRG2557 from the Estonian Research Council and TK210 from the Estonian Ministry of Education and Research.)