On March 6, 2026 the on-line course (MOOC) LC-MS Method Validation offered by University of Tartu finished successfully.
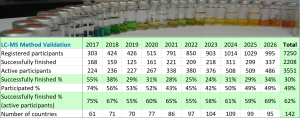
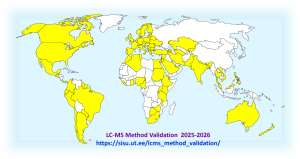
Eventually, altogether 995 people registered from 95 countries. Roughly half of them, 486 participants, actually started the course (i.e. tried at least one graded test at least once), and out of them, 337 successfully completed the course. The overall completion rate was 34%. The completion rate of participants who started the studies was 69%. The completion rates of active participants (i.e. who started the course) seem to have stabilised at around 60% and have increased during the current run. In any case, there is some food for thought for us on how we could improve the participation rate…
At the same time, those participants who actually took part in the course were very active and asked lots of questions. The questions were often very much to the point and addressed things that are really important to analysts in their everyday work. The course has several forums (general and by topic). During the course period, the overall number of forum posts was around 200 (overall number of posts, both from participants and from teachers), and the forums are still active, and posts are still coming in.
This active participation made teaching this MOOC a great experience also for us, the teachers. The discussion threads gave a lot of added value to the course.
We want to thank all participants for helping to make this course a success!
We plan to repeat this course again in Autumn 2026.