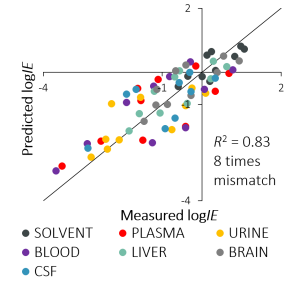 It is a pleasure to see the real-life application of one’s research. Even more so if discoveries are made during “routine” work. We aimed to develop and validate a UHPLC-MS/MS assay suitable for the quantifying simultaneously two important cardiovascular drugs – milrinone and dobutamine. Dobutamine has a half-life of 2 minutes and unfortunately also undergoes rapid degradation in plasma samples, which we managed to get under control by the addition of ascorbic acid (vitamin C). The study aims to determine the pharmacokinetics of these drugs in neonates and paediatric patients, which restricted the sample amount for the assay. Nonetheless, we achieved the full validation using only 20 µL of human plasma for the analysis, while still reaching the required lowest limit of quantification of 1 ng/mL. This was facilitated by the use of ammonium fluoride as an eluent additive, which provided a significant signal enhancement in positive electrospray ionisation (an effect not entirely understood yet). This collaborative study was recently published in Clinical Mass Spectrometry.
It is a pleasure to see the real-life application of one’s research. Even more so if discoveries are made during “routine” work. We aimed to develop and validate a UHPLC-MS/MS assay suitable for the quantifying simultaneously two important cardiovascular drugs – milrinone and dobutamine. Dobutamine has a half-life of 2 minutes and unfortunately also undergoes rapid degradation in plasma samples, which we managed to get under control by the addition of ascorbic acid (vitamin C). The study aims to determine the pharmacokinetics of these drugs in neonates and paediatric patients, which restricted the sample amount for the assay. Nonetheless, we achieved the full validation using only 20 µL of human plasma for the analysis, while still reaching the required lowest limit of quantification of 1 ng/mL. This was facilitated by the use of ammonium fluoride as an eluent additive, which provided a significant signal enhancement in positive electrospray ionisation (an effect not entirely understood yet). This collaborative study was recently published in Clinical Mass Spectrometry.
New Publication: Determination of pKa values of fluorocompounds in water using 19F NMR
 Our group is well-known for pKa measurements in different solvents. So far, mostly UV-Vis spectrophotometric method has been used. Recently Elisabeth Parman (on the photo) from our group has been developing a method for measuring pKa values of fluorocompounds, using 19F NMR. NMR measurements to determine pKa values have several advantages compared to UV-Vis spectrophotometry. For example, accurate concentrations are not needed, the method is much more tolerant to impurities and several compounds can be measured simultaneously. The newly developed method has now been published in the Journal of Physical Organic Chemistry: Determination of pKa values of fluorocompounds in water using 19F NMR by E. Parman, L. Toom, S. Selberg and I. Leito.
Our group is well-known for pKa measurements in different solvents. So far, mostly UV-Vis spectrophotometric method has been used. Recently Elisabeth Parman (on the photo) from our group has been developing a method for measuring pKa values of fluorocompounds, using 19F NMR. NMR measurements to determine pKa values have several advantages compared to UV-Vis spectrophotometry. For example, accurate concentrations are not needed, the method is much more tolerant to impurities and several compounds can be measured simultaneously. The newly developed method has now been published in the Journal of Physical Organic Chemistry: Determination of pKa values of fluorocompounds in water using 19F NMR by E. Parman, L. Toom, S. Selberg and I. Leito.
In this work, the developed method was applied to 19 fluorocompounds – fluorinated phenols, carboxylic acids and sulfonamides, hexafluoroisopropanol (HFIP), perfluoropinacol, etc – with the aim of determining their pKa values in water, in the range from about 3.5 to 10. Several advantages of the 19F NMR method were demonstrated in pKa measurement. In particular, using multiple relative measurements against several reference compounds with known acidities it is possible to obtain highly reliable pKa values. Elisabeth convincingly demonstrated that the literature pKa values of two of the investigated compounds were inaccurate and provided more reliable values for them.
New type of pKa – biphasic pKa in octanol water system
 It is not frequent that a new quantitative measure of some physicochemical characteristic is introduced. Recently our group managed to do exactly that by introducing a novel approach – termed as biphasic pKa values – of measuring the acidity/basicity of lipophilic compounds embedded in water-immiscible media (membranes or solutions) at equilibrium with aqueous solution.
It is not frequent that a new quantitative measure of some physicochemical characteristic is introduced. Recently our group managed to do exactly that by introducing a novel approach – termed as biphasic pKa values – of measuring the acidity/basicity of lipophilic compounds embedded in water-immiscible media (membranes or solutions) at equilibrium with aqueous solution.
In this approach, the H+ ion activity is determined in the aqueous phase, while the concentration ratio of protonated and deprotonated forms of acid/base is measured in the lipophilic phase with any suitable technique. The approach has been demonstrated on the example of biphasic pKa values of some lipophilic indicator bases in the octanol: water system, measured using UV-Vis spectrophotometry. The counterion identity and concentration, as well as the overall ionic strength in the aqueous phase, are included in the standard state definition.
This work has been published S. Selberg, S. Tshepelevitsh, I. Leito Croat. Chem. Acta 2018, 91, (4) and will form a part of the PhD thesis of Sigrid Selberg.
Biphasic pKa values are envisaged to be useful for characterizing lipophilic indicators, synthetic receptors, phase transfer catalysts and other implements that are used in hydrophobic media (e.g. in sensor membranes) in contact with water.
This work is part of a larger endeavor – to promote a wider usage the unified pH scale (pHabs) by the research and technology communities, which is currently in progress via the UnipHied.
 The UnipHied project is funded from the EMPIR programme (project 17FUN09) co-financed by the Participating States and from the European Union’s Horizon 2020 research and innovation programme.
The UnipHied project is funded from the EMPIR programme (project 17FUN09) co-financed by the Participating States and from the European Union’s Horizon 2020 research and innovation programme.
(On photo: Sigrid Selberg)
Optimization of flow splitting and make‐up flow conditions in liquid chromatography/electrospray ionization mass spectrometry

Recently a study in Rapid Communications in Mass Spectrometry was published. In this study, we optimized the actual split ratio and make-up flow composition in LC/ESI/MS analysis to increase the signal in radio detector and enhance the sensitivity of electrospray ionization.
High methanol content increases electrospray ionization efficiency
We studied make-up flow composition with a set of 20 pharmaceuticals covering 21 different make-up flow compositions. We studied methanol, acetonitrile, isopropanol and acetone as organic modifiers. As we studied positive mode the acidic additives under investigation were formic acid, oxalic acid and propionic acid. DMSO and sulfolane as additives were studied as well. The Methanol/10 mM formic acid in water (90/10) proved to be the best make‐up flow composition in relation to the average sensitivity obtained. Stronger acidic conditions using oxalic acid or higher formic acid concentrations had a clear positive effect on the sensitivity of compounds with low ionization efficiency.
Split-ratios remain stable over main part of the gradient
 The second part of this study was testing and monitoring different split ratios (1:10, 1:100 and 1:250) with different splitters (Alliance flow‐splitter kit and homemade T-piece splitter). To monitor the actual split ratio haloperidol solution was added with infusion pump post-column while a haloperidol-d4 solution was added as a make‐up flow by the ISM pump after the splitter. The tested split ratios were relatively stable over the main part of the gradient but showed some variation at very low and very high organic conditions. Differences were larger with methanol compared with acetonitrile containing solvent compositions and when applied without a column or with very long connecting tubing.
The second part of this study was testing and monitoring different split ratios (1:10, 1:100 and 1:250) with different splitters (Alliance flow‐splitter kit and homemade T-piece splitter). To monitor the actual split ratio haloperidol solution was added with infusion pump post-column while a haloperidol-d4 solution was added as a make‐up flow by the ISM pump after the splitter. The tested split ratios were relatively stable over the main part of the gradient but showed some variation at very low and very high organic conditions. Differences were larger with methanol compared with acetonitrile containing solvent compositions and when applied without a column or with very long connecting tubing.
I thank Janssen Pharmaceutica and especially Dr Filip Cuyckens for the internship position for letting to gain experience in an industry setting and study these effects. Additionally, I thank Erasmus+ mobility and Smart specialization doctoral stipend for funding my stay.
Comparing Electrospray Ionization Efficiencies of Derivatized and Non-Derivatized compounds
 Main concern while measuring ionization efficiency (IE) of derivatized compounds is that derivatized compounds are in complex mixtures. To overcome this problem, we developed a chromatographic method to separate these compounds and measure their ionization efficiencies.
Main concern while measuring ionization efficiency (IE) of derivatized compounds is that derivatized compounds are in complex mixtures. To overcome this problem, we developed a chromatographic method to separate these compounds and measure their ionization efficiencies.
If ionization efficiencies of derivatized compounds are measured without chromatographic separation in a complex mixture, which includes analytes, derivatization reagents and different by-products, then matrix effects could occur. This problem can be solved if we first separate these compounds from each other and then measure ionization efficiencies.
Chromatographic method
To separate different compounds, we used a simple chromatographic gradient elution method. One might think, that if compounds have different retention time, then they also elute at different organic phase percentages which obviously affects measured ionization efficiency values. The effect is not so significant, but we could manage to model it and take it into account to level all newly measured ionization efficiency values to a previously measured scale.
logIE measurements
We carried out ionization efficiency measurements with liquid chromatography electrospray ionization mass spectrometry (LC/ESI/MS) and constructed IE scales with a calibration curve for compounds with and without derivatization reagent diethyl ethoxymethylenemalonate (DEEMM). Additionally, we investigated eluent composition effects on ionization. Measured compounds were mainly amino acids but also included some biogenic amines.
Results
We saw, that
- Derivatization increases IE for most of the compounds (by an average of 0.9 and up to 2 – 2.5 logIE units) and derivatized compounds have more similar logIE values than their underivatized variants.
- Measurement of derivatized compounds is possible from the mixture when using chromatographic separation technique.
- It was also noted, that using chromatographic separation instead of flow injection mode tends to slightly increase ionization efficiency.
We managed to link all our results with existing ionization efficiency scale and therefore widen the scale and get comparable logIE values for measured derivatized compounds.
More information could be found in an article published recently at Journal of Mass Spectrometry.
Milestone in catalysis research: approaching sub-ppm-level asymmetric organocatalysis
 Recently our group had the pleasure of participating in a cutting-edge endeavor: characterizing the acidity of some extremely efficient strongly acidic organocatalysts. In the case of the Mukaiyama aldol reaction, the best of them (1) worked at low ppm to sub-ppm level, (2) gave excellent yields and (3) high enantiomeric selectivity as well as (4) turnover numbers (TON numbers) of hundreds of thousands.
Recently our group had the pleasure of participating in a cutting-edge endeavor: characterizing the acidity of some extremely efficient strongly acidic organocatalysts. In the case of the Mukaiyama aldol reaction, the best of them (1) worked at low ppm to sub-ppm level, (2) gave excellent yields and (3) high enantiomeric selectivity as well as (4) turnover numbers (TON numbers) of hundreds of thousands.
The extent to which these four features occurred together in the same catalyst was so remarkable that the results were published in one of the most prestigious journals in chemical sciences: Nature Chemistry 2018, 10, 888-894.
The extremely demanding acidity measurements were performed by Dr Karl Kaupmees. The whole research was led by the group of professor Benjamin List – a worldwide known guru in the field of strongly acidic catalysts working at the Max-Planck-Institut für Kohlenforschung.
These results are expected to open new avenues in the development of powerful new organocatalysts.
(Photo by Andres Tennus: Karl doing acidity measurements in a glovebox under anhydrous conditions)
Prediction of ionization efficiencies in biological matrices
In the recent years, we have made significant progress in ionization efficiency studies. In our previous publications, we have shown that our approach can be applied in both electrospray positive and negative mode (we have recently also enabled comparing the logIE values measured in both ESI modes numerically), that our approach is transferable between different instruments with various ESI source geometries from all major mass spectrometric systems vendors, and that it is also transferable between different eluent compositions.
The logIE approach is already applicable in nice clean matrices such as neat solvents. We wanted to see if our approach also works with more difficult matrices, such as bodily fluids and tissues. The results were recently published in Analytica Chimica Acta.
In this study, we took a representative set of 10 compounds, including drugs, e.g., naproxen and lincomycin. As matrices, we used blood, plasma, urine, cerebrospinal fluid, brain and liver tissue homogenates and neat solvent to compare with. We carried out a simple and robust sample pretreatment of protein precipitation. We measured the IEs in a worst-case scenario in flow injection mode without any chromatographic separation.
As with different instrumental setups and solvents, the IEs vary between biological matrices, but the order of the compounds remains roughly the same. These variations between different matrices and variations between a certain biological matrix and neat solvent demonstrate that matrix affects ionization efficiencies and also the prediction models of ionization efficiencies. This, in turn, shows that matrix affects the importance of properties of compounds in the prediction model.
Even though the effects are big and matrix effect is strong we were happy to see that the correlations between IEs measured in the neat solvent and IEs measured in a biological matrix are in good correlation (R2 from 0.7 to 0.9). These good correlations were a promising start to predict IEs in biological matrices similarly to previous predictions in the neat solvent. The most accurate model was obtained for the solvent with a mismatch of 2 times which was also expected since it is the cleanest matrix. But also for liver and brain tissues the mismatch of the model is only 3-fold.
The correlation between predicted and calculated IEs is good with the average mismatch over all biological matrices of 8 times. This means that the accuracy of standard substance free quantitation has been improved by more than an order of magnitude for the set of compounds used in this study.
More details can be found in the paper published in Analytica Chimica Acta. Piia Liigand also gave a talk on the topic in ASMS which can be found here. More papers by our group on the topic of ionization efficiencies can be found here.
Ancient men and women had different menus!
 A new paper on ancient dietary practices was recently published by our group (led by Dr. Ester Oras) in the Journal of Archaeological Science: “Social food here and hereafter: Multiproxy analysis of gender-specific food consumption in conversion period inhumation cemetery at Kukruse, NE-Estonia”.
A new paper on ancient dietary practices was recently published by our group (led by Dr. Ester Oras) in the Journal of Archaeological Science: “Social food here and hereafter: Multiproxy analysis of gender-specific food consumption in conversion period inhumation cemetery at Kukruse, NE-Estonia”.
We demonstrated the fruitfulness of multiproxy dietary analysis combining plant microfossil, human bone stable isotope and pottery related organic residue analysis. The results reveal that even 800 years ago men and women had different dietary habits: men preferred fish and higher trophic level terrestrial animals (e.g. pork), whilst women declined towards ruminant carcass (a nice steak!) and dairy products.
The paper is one of the few of its kind illustrating ancient food consumption as a highly social phenomenon, and setting an example for microscale dietary analysis in the future.
Is it possible to use data below the limit of quantitation in pharmacokinetic studies?
 Results below the limit of quantification (BLQ) are generally not reported or reported without explicit numbers, leaving pharmacokinetic (PK) and pharmacodynamic (PD) studies wishing for more information to improve their model parameters. But the laboratory cannot release actual values BLQ since the quality of that data has never been assessed.
Results below the limit of quantification (BLQ) are generally not reported or reported without explicit numbers, leaving pharmacokinetic (PK) and pharmacodynamic (PD) studies wishing for more information to improve their model parameters. But the laboratory cannot release actual values BLQ since the quality of that data has never been assessed.
Possible solutions to this unfortunate situation were investigated in a recent perspective article led by our group and published in Bioanalysis. The article titled: “Utilization of data below the analytical limit of quantitation in pharmacokinetic analysis and modelling: promoting interdisciplinary debate”, expanse the so far used statistical salvage of information, by an experimental addition to investigate the quality of data BLQ.
By directing this article towards the pharmacometrician, the analytical scientist and the regulatory personnel, we hope to encourage an inter-disciplinary discussion to improve the situation by finding ways to use BLQ data in PK/PD studies, in order to enhance the quality of the obtained pharmacokinetic models. Several ways were proposed for moving forward, in particular improving/modifying method validation guidelines for enabling to use BLQ data and leaving the decision regarding whether and how to incorporate the BLQ data into a PK/PD model to the data analyst and not the analytical chemist.
Pan-European Network of Fundamental pH Research: UnipHied
 Initiated by our group, the pan-European research network of fundamental pH Research UnipHied started in May 2018.
Initiated by our group, the pan-European research network of fundamental pH Research UnipHied started in May 2018.
Why is such network needed? As of now, it is not possible to compare pH values of solutions made in different solvents, as every solvent has its own pH scale. This situation is highly unfortunate, since it causes confusion and inaccuracies into many fields, extending far beyond the specific field of acid-base chemistry. Examples are industrial catalytic processes, food chemistry, liquid chromatograpy, etc. The central goal of UnipHied is to overcome this situation by putting the new theoretical concept of the recently introduced unified pHabs scale on a metrologically well-founded basis into practice.
The most important specific objectives of UnipHied are (1) to develop and validate a reliable and universally applicable measurement procedure that enables the measurement of pHabs; (2) to create a reliable method for the experimental or computational evaluation of the liquid junction potential between aqueous and non-aqueous solutions; (3) to develop a coherent and validated suite of calibration standards for standardizing routine measurement systems in terms of pHabs values for a variety of widespread systems (e.g., industrial mixtures, soils/waters, food products, biomaterials).
The first version of the pHabs measurement procedure has been created by Agnes Heering (Suu) in the framework of her PhD thesis. The main experimental difficulty is evaluation of the liquid junction potential (LJP), which will be thoroughly addressed by UnipHied. The first important steps towards this goal have very recently been made and published as two back-to-back papers: Angew. Chem. Int. Ed. 2018, 57, 2344–2347 and Angew. Chem. Int. Ed. 2018, 57, 2348–2352
The key achievement described in the papers is finding an ionic liquid, namely [N2225][NTf2], that can be used as salt bridge electrolyte and has such properties that two out of three main sources of LJP are eliminated.
The partners of the UnipHied network are LNE (France, coordinator), BFKH (Hungary), CMI (Czech Republic), DFM (Denmark), IPQ (Portugal), PTB (Germany), SYKE (Finland), TÜBITAK-UME (Turkey), Freiburg University (Germany), ANBSensors (United Kingdom), FCiencias.ID (Portugal), UT (Estonia).
 UnipHied is funded from the EMPIR programme (project 17FUN09) co-financed by the Participating States and from the European Union’s Horizon 2020 research and innovation programme.
UnipHied is funded from the EMPIR programme (project 17FUN09) co-financed by the Participating States and from the European Union’s Horizon 2020 research and innovation programme.